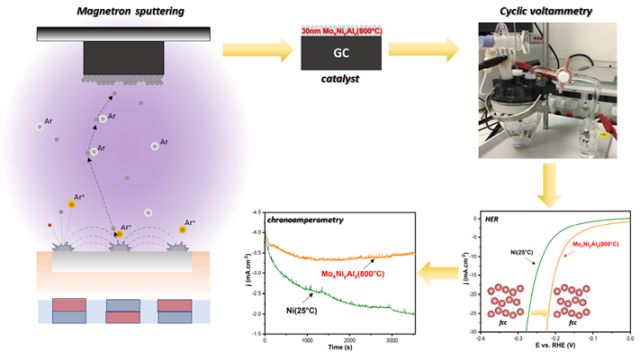
In the present article, we studied the catalytic activity of magnetron-sputtered MoxNiyXz (where X = Al, Co, Cr, Cu, Fe, W) thin films for the hydrogen evolution reaction (HER) in 1 M NaOH. Thin-film electrocatalysts were prepared at 25 °C and 800 °C and characterized by SEM-EDX and XRD analyses. The Mo80Ni5Al15(800 °C) electrocatalyst demonstrated the best HER overpotential (-181 mV) among all tested ternary systems. XRD analysis revealed that only the Mo80Ni5Al15 thin films prepared at 800 °C exhibited the fcc phase, while less active compositions contained only the bcc phase. Electrochemical impedance spectroscopy results were consistent with cyclic voltammetry measurements. Chronoamperometry showed that the HER activity of Mo80Ni5Al15(800 °C) remained stable after a slight initial drop, whereas the activity of Ni catalysts nearly halved over the same time frame. These findings suggest that Mo80Ni5Al15(800 °C) could serve as a promising alternative to industrial Ni-based catalysts for alkaline water electrolyzers.